Sea cucumbers- Inside out
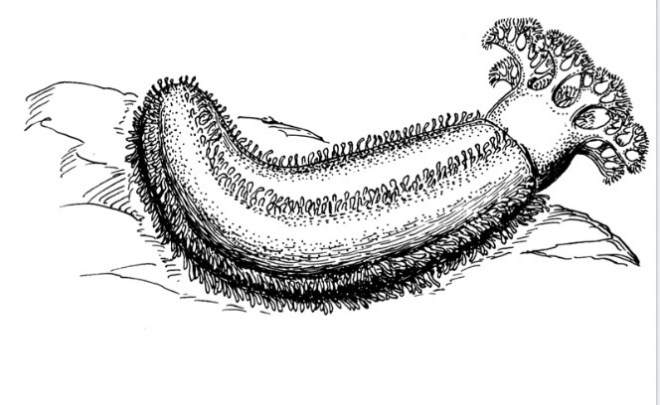
“If in doubt, gross them out!”
It’s a phrase I’ve said many times at my aquarium day job, usually while explaining my techniques for engaging with members of the public. And I stand by it to this day, because there are many creatures of the sea (and beyond) who resort to rather…..
Read more
Post of the month

21st May 2026
Happy World Cultural Diversity Day 🗺️
With their incredible size and graceful nature, whales have left their mark on many cultures. Including, but not limited to:
- Japanese swordmaster Miyamoto Musashi attacking a giant whale.
- The thunderbird, or Q’wati-tha in the language of the Makah Tribe from Washingon, USA, catching a whale in its claws.
- The story of Jonah and the whale, as told in both the Hebrew bible and the Quran.
For more bitesize content and ocean stories why not follow ‘Our world under the waves’ on…
🌊 Facebook 🌊 Bluesky 🌊 Instagram
Latest on the horizon
🌊 New article out now! Discover the ever gross out secrets of a sea cucumber!
🌊 Original novel Shark on a train awaiting publication.
🌊 Save the date
💧 8th June- World Ocean Day
💧 11th June- Jacques Cousteau’s Birthday
💧 26th June- International Angel Shark Day














